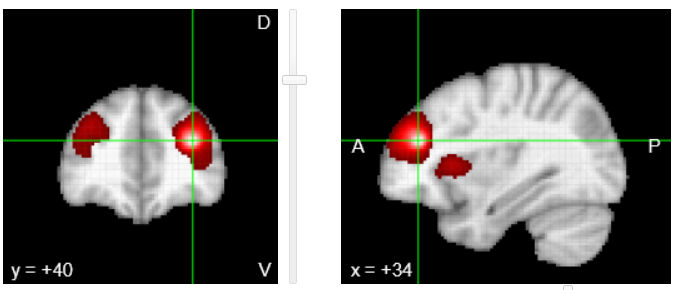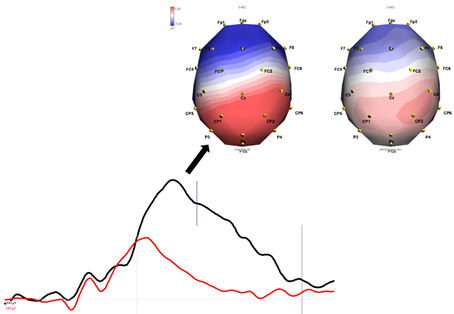
We will cover a variety of foundational and contemporary issues in human neuroscience, and will engage in discussion of scientific methods and techniques used to investigate the relationship between the function of brain systems, behavior, and cognition. The course will primarily emphasize functional neuroanatomy, higher cognitive and emotional function (cognitive and affective neuroscience), the neurobehavioral underpinnings of pathology in humans (clinical neuroscience), and an emerging field known as “positive” neuroscience. Laboratory sections will supplement lecture and reading topics with demonstrations and experience with neuropsychological, cognitive neuroscience, and psychophysiological research techniques. Both the lecture and laboratory portions of this course strongly stress experimental design, data analysis, and scientific reasoning. However, we also get to play with brains. Brains are pretty rad.
- Teacher: Todd Watson
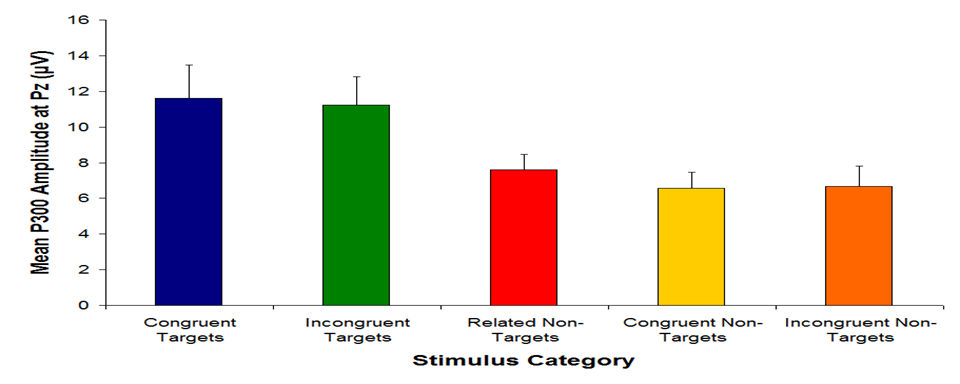
You will gain an understanding of descriptive statistics (used to summarize data) and basic inferential statistics (used to test hypotheses and draw conclusions) using the “classical” or “frequentist” model. You will learn to apply the principles learned during the course to not only to better understand psychology and other fields of scientific research, but also to understand how statistics are used (and sometimes abused) in daily life. Topics will include the properties of numerical distributions, measures of central tendency, variability, correlation/bivariate regression, t-tests, analysis of variance (ANOVA), and a tiny (and I mean tiny) bit on nonparametric statistical techniques. I also promise that all these words will make sense by the end of the semester. In addition, you will gain experience using statistical software (JASP) to analyze and display research data. Finally, you will gain experience in presenting research findings in American Psychological Association (APA) format.
Important Note: You can’t earn credit for BOTH Psy 200 and AP/IB statistics.
Important Note: You can’t earn credit for BOTH Psy 200 and AP/IB statistics.
- Teacher: Todd Watson
Numbers teaches students to interpret quantitative, information presented in various forms and, contexts; to understand the logical structure of, quantitative arguments; and to use quantitative, models, theories, and data to simplify, explain,, and make predictions. Specific content and topics, will vary with instructors.
- Teacher: Diana Leonard